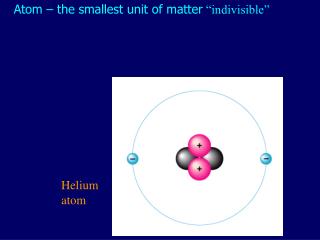
The food you eat, the house you live in and the rocks and soil you walk on all expose you to low levels of radioactivity. The average American receives a radiation dose of about 620 millirem every year. The radioactivity born inside your body is only a fraction of the radiation you naturally (and harmlessly) come in contact with on an everyday basis. But it’s not long before these positrons bump into your electrons and annihilate into radiation in the form of gamma rays. The average human produces more than 4000 positrons per day, about 180 per hour. When Potassium-40 decays, it releases a positron, the electron’s antimatter twin, so you also contain a small amount of antimatter. They are incorporated into your molecules and eventually decay and produce radiation in your body. You emit radiation because many of the foods you eat, the beverages you drink and even the air you breathe contain radionuclides such as Potassium-40 and Carbon-14. Your radiation dose level can go up by one or two millirem for every eight hours you spend sleeping next to your similarly radioactive loved one. That’s the same amount of radiation you’d be exposed to from having four chest X-rays. You receive an annual 40-millirem dose from the natural radioactivity originating inside of you. Your body is a small-scale mine of radioactive particles. Scientists believe that almost all of your body’s mass comes from the kinetic energy of the quarks and the binding energy of the gluons. If your mass doesn’t come from the masses of these particles, where does it come from? Energy. Gluons, carriers of the strong nuclear force that holds these quarks together, are completely massless. The mass of the quarks, which comes from their interaction with the Higgs field, accounts for just a few percent of the mass of a proton or neutron. The protons and neutrons inside of an atom’s nucleus are each made up of three quarks. If we lost all the dead space inside our atoms, we would each be able to fit into a particle of lead dust, and the entire human race would fit into the volume of a sugar cube.Īs you might guess, these spaced-out particles make up only a tiny portion of your mass. If the nucleus were the size of a peanut, the atom would be about the size of a baseball stadium. Nuclei are around 100,000 times smaller than the atoms they’re housed in. The size of an atom is governed by the average location of its electrons. The very heavy elements in you were made in exploding stars. While most of the cells in your body regenerate every seven to 15 years, many of the particles that make up those cells have actually existed for millions of millennia. The hydrogen atoms in you were produced in the big bang, and the carbon, nitrogen and oxygen atoms were made in burning stars. In comparison, the top quark weighs 57,500 times more than the up quark.About 99 percent of your body is made up of atoms of hydrogen, carbon, nitrogen and oxygen. You also contain much smaller amounts of the other elements that are essential for life. Up and down quarks make up protons and neutrons, and they weigh 3 million and 5 million electron volts, respectively. Scientists have found six types of quarks: up, down, strange, charm, bottom and top.

Quarks are another fundamental particle that, as far as we know, cannot be broken down into more parts. For comparison, a typical proton in the nucleus of a typical atom weighs 938 million electron volts, or 1.67 × 10^-27 kg, he said.Ĭonversely, the largest (in terms of mass) fundamental particle we know of is a particle called a top quark, measuring a whopping 172.5 billion electron volts, according to Lincoln. To figure out the mass of a subatomic particle, then, you'd use Albert Einstein's famous equation E=mc^2 to get the mass (m) in kilograms.Īn electron weighs 511,000 electron volts, which is equivalent to 9.11 x 10^-31 kilograms, according to Lincoln. To simplify things, physicists use a set of units whereby the speed of light is 1. One electron volt is equivalent to about 1.6x10^-19 joules.
Technically, the unit is eV/c^2, in which c is the speed of light.
Physicists use electron volts (eV) to measure the mass of subatomic particles, Lincoln said. The size of particles can be measured by investigating their mass or their physical size.
